|
|
Issue
33: Summer 2003
Table of Contents
Boycott news 33 - the latest on the Nestlé boycottIBFAN 2004 Breastfeeding
calendar now available in the on-line Virtual
Shop.
|
|||||||||||||||||||||||||||||
A mother
complains to her local Trading Standards office about what
she thinks is an infant formula advertisement. This simple
action sparked an 8-day trial of a US pharmaceutical company
that goes to the heart of concerns about the impact of trade
on health, and will have enormous significance for mothers
and infants. Wyeth took on Birmingham Trading Standards’ Legal Unit, with an attack on the UK Government’s right to regulate the marketing of baby milks. The case suggests that similar advertisements (many of which were examined by the court) are illegal. |
The advertisement at the centre of the case. Prima magazine, July 2001. |
The judgment will send a message to the whole industry that it can no longer be complacent about the many other current marketing practices that violate the UK Law such as irresponsible labelling and direct contact to mothers (through Carelines etc).
Judge Ross said:
“The Defendants have deliberately ‘crossed the line’ in an effort to advertise direct to a vulnerable section of society. This is a cynical and deliberate breach of the regulations.”
Noting that Graham Crawford, SMA Director and Chair of the UK Infant and Dietetic Foods Association (IDFA), had ignored the advice of an in-house lawyer that the article was probably a breach of the law, (Wyeth claimed that the lawyer was new to the company), Mr Ross said:
“It is clear to me that the defendant knew very well that the article could be perceived by the public as an advert for SMA Gold... They were prepared to ignore the advice of their legal department and took the risk of prosecution.”
The European point
Wyeth/SMA brought in Professor Alan Lucas as an expert witness to back up its claim that the ‘information piece’ was not harmful and did not ‘downgrade the distinction between breastfeeding and formula.’ The company intended to use this to prove that the UK’s advertising ban is unnecessary on health grounds and that it ‘fetters the free movement of goods,’ stopping companies such as Nestlé, entering the market. |
“As somebody who has spent
his professional life concerned with ways of improving childrens'
health through improving nutrition and diet and as somebody
who sees this being eroded by attempts to violate the WHO Code
I applaud Trading Standards’ prosecution of SMA. I offer
my thanks and congratulations, but more importantly please,
on their behalf, accept the thanks of children born and yet
to be born who will benefit from your successful case.” |
The Company said that Germany permits advertising of infant formula but attempted to exclude evidence that many other EU countries, such as France, the Netherlands, Belgium, Denmark and Luxembourg, have banned it, as specifically permitted under European legislation.
| The judgment should strengthen the case for bringing the UK Law into line with the International Code and Resolutions, which are policies agreed by the World Health Assembly, the world’s highest health policy setting body. If the case had been lost on the European Point (or does so on a possible appeal), the UK would have been under pressure to ensure that its legislation is no stricter than the weakest of any other country in Europe. | Wyeth/SMA facts: US pharmaceutical company (formerly called American Home Products) Annual sales 2002: $14.6 billion. 2nd largest baby food company and Code violator in the world. In 1983, Wyeth led the industry in forming a weak UK voluntary code, effectively delaying UK legislation for years. When the EU Directives were adopted in 1991, Wyeth changed its name to SMA Nutrition to match the UK brand and get round the advertising restrictions. The Wyeth advertisements and slogans used in the UK are found by IBFAN all over the world. |
Judge Ross said:
“In my view the manufacturers are playing on a ‘level playing field’...It is clear that it is important to uphold the law of the land in the public interest bearing in mind the stability in our society.”
Mr Ross said that Graham Crawford had not excercised ‘due diligence’ and had been “extra-ordinarily evasive throughout his cross-examination. His expertise was rather less than he wanted me to believe.” In mitigation SMA claimed that it had been ‘misled’ by the activities of other companies.
|
Above. Ms Andrews, SMA defence lawyer, claiming that photographing SMA Director, Mr Crawford (above left) outside the court was an infringement of his personal privacy. Interesting, considering her treatment of one of the civilian witnesses. When the prosecution asked about the relevance of her questions, the Judge commented: “She is attacking the character of the witness in an underhand way, attempting to ridicule her.” |
Not so shy campaigners who came to hear the verdict: Mike Brady, Magda Sachs, Patti Rundall and Peter Greaves. |
What the case was about
The International Code of Marketing of Breastmilk Substitutes bans advertising of breastmilk substitutes, but the UK Infant Formula and Follow On Formula Regulations 1995 allow advertisements for infant formula for babies up to six months to be published or displayed through the healthcare system, in a scientific publication, in a publication not widely available to the public, or for the purposes of trade before retail. The six advertisements placed by SMA violated the Law because they were in magazines on sale in shops. The EU Directive which gave rise to the UK law specificially permits the banning of advertising.
Despite the UK Government’s consistent support for the Resolutions passed at the World Health Assembly, the UK has yet to ensure that its law meets the minimum requirements which will protect the rights of infants in this country.
Write to Melanie Johnson, Minister for Public Health, to ask for the law to be brought into line with the International Code and Resolutions. Department of Health, Richmond House, 79 Whitehall, London, SW1A 2NS. http://www.doh.gov.uk/index.htm
For copies of the legislation, its amendments, a commentary on how it should be improved, how to monitor and suggestions for what you can do to help, go to: http://www.babyfeedinglawgroup.org.uk/
There is no central register of complaints made to Trading Standards so please remember to copy correspondence to us.
Prof. Lucas sits on SMA team
Wyeth/SMA brought in as an expert witness for the defence, Alan Lucas, Professor of Paediatric Nutrition at the Institute of Child Health, London and world renowned researcher. His selective use of information during this case has shocked many campaigning for infant health and will inevitably affect how he is viewed.
Prof Lucas sat with Wyeth’s defence team throughout the trial, submitting an expert report which SMA used in its attack on the UK ban on advertising. With his extensive knowledge of the benefits of breastfeeding, which he acknowledged several times, he walked a fine line. He asserted that since the majority of babies in the UK are bottle fed it was important to work closely with companies to ensure that babies receive the best alternative possible and mothers are properly informed. He said he wanted to promote breastfeeding, and to prevent “reckless advertising” and any “downgrading” of breastfeeding. However, he ignored the basis on which the International Code is adopted as a universal recommendation and a minimum requirement for all countries, taking the position that the SMA advert was an industry standard and was a reasonable compromise for “the West.” Several times he said he was not aware of any data that demonstrates that an information piece, such as the one in question, is harmful.
Prof Lucas said that GPs and primary healthcare workers have poor understanding of infant nutrition and that companies hold important information that mothers need to know. He referred to company-operated Carelines (which Baby Milk Action believes to be illegal) as a possible source of information for mothers. These points were not relevant to the question “is this an advert or not?”, which the judge kept stressing, but they may be relevant if Wyeth takes the case to appeal.
Unfortunately for the defence, while attempting to prove that Wyeth was no worse than any other company, Prof Lucas substantially undermined the company’s main argument that the piece in question was not an advertisement. He said it did indeed point to a particular product - as did many of the 15 advertisements by other companies which were examined by the court. He claimed that the mention of ingredients, even without a logo, would be enough to identify a product, saying, “Why would they talk about LCPs if they didn’t have a product with it in?”
Prof Lucas claimed that he was ‘neutral’ and there to ‘help both sides’. However, several times the prosecution Barrister, Richard Barraclough QC, reminded him that as an impartial expert witness he should ensure that the court is made aware of contrary opinions, suggesting that Prof Lucas had allowed his judgement to be clouded by his own treatment. Prof. Lucas had termed one breastfeeding support group as ‘extreme’ because of its rules on conflicts of interest. Prof Lucas apologised for this and for leaving out key information from his report, but vehemently denied being ‘selective’.
Prof Lucas listed his numerous achievements, including his many clinical posts, credits, and 320 articles, reviews and books on paediatrics. He stated that his centre holds the best data on the use of human milk for sick infants, milk banking, the impact of infant feeding on long-term health and that his research is used by breastfeeding advocacy groups.
Surprisingly, Prof Lucas said he did not know of the UK Government targets for breastfeeding, but had drafted a letter which he said he would probably be sending to the Department of Health, asking it to reconsider its recommendation on 6 months exclusive breastfeeding.
He said he was unaware that the Committee on the Convention on the Rights of the Child had called on the UK Government to implement the International Code. He acknowledged that his original report setting out advertising regulations in other countries wasn’t balanced.
The defence asked Prof Lucas to talk about “possible adverse effects of breastfeeding,” referring to his own research. One controversial study, whose methodology has been widely challenged, carried the inference that breastfeeding followed “by a western diet” could increase the risk of heart disease. (Ref. 1) The study was published in the British Medical Journal with two press releases, so generated worldwide misleading publicity (including in India) suggesting that breastfeeding may cause heart disease, despite the fact that the data did not confirm this conclusion (see report in Update 29).
As evidence of his links with breastfeeding advocacy groups he cited lectures to La Leche League and claimed that one of his chief scientists is an advisor to the National Childbirth Trust. (NCT are baffled by this claim.)
Prof Lucas acknowledged extensive ‘involvement’ with the baby food industry, saying that he held several advisory posts in the EU, US and in the UK, including the Chair of an Advisory group to the industry lobbying body, INFORM. He said his role is to advise industry on how to properly position themselves in the UK. He did not mention funding, until questioned by the prosecution. To us, following the case, he has confirmed that until recently, nearly 70% of his funding came from baby food manufacturers (it is unclear whether this is his personal or institutional research income). He told us his research is now predominently publicly funded. In court he denied the suggestion that he was part-funded by SMA, claiming that their research relationship was “symbiotic.”
He told us he had not decided whether to accept his fee for his work relating to the court case for Wyeth/SMA.
(Ref. 1) Leeson et al, (2001) Duration of breastfeeding and arterial distensibility in early adult life: population based study, BMJ, Vol. 322, (643-647). The 42 responses to this paper can be found on http://bmj.com/cgi/eletters/322/7287/643
Does commercial promotion affect breastfeeding rates?
SMA’s expert witness Prof Alan Lucas claimed he was unaware of any data that demonstrates that commercial material, such as the SMA piece, are harmful. However, a simple search on an electronic search engine such as Medline, reveals over 200 articles addressing the widespread concern about the impact of commercial promotion especially within health care settings. Here are just a few.
Office prenatal formula advertising and its effect on breast-feeding patterns. Howard C et al. Obstetrics and Gynaecology Vol 5, No 2, Feb 2000 p296-303 This study of 547 pregnant women, compares the effect of formula company-produced materials about infant feeding to breast-feeding promotion materials without formula advertising on breast-feeding initiation and duration. Although breast-feeding initiation and long-term duration were not affected, exposure to formula promotion materials increased significantly breast-feeding cessation in the first 2 weeks. Additionally, among women with uncertain goals or breast-feeding goals of 12 weeks or less, exclusive, full, and overall breastfeeding duration were shortened. The study concludes that formula promotion products should be eliminated from prenatal settings.
Evidence for the 10 Steps to successful breastfeeding, Tables 1.1, and 6.4 and 6.5. WHO Geneva 1998 This (and many other useful documents) can be downloaded from WHO’s website: http://www.who.int/child-adolescent-health/publications/NUTRITION/WHO_CHD_98.9.htm
The influence
of Infant Food Advertising on infant feeding practices in St
Vincent, International Journal of Health Services Vol
12 No 1 1982 p 53 to 75.
A Social Science Perspective on Gifts to Physicians
from industry, Dana J et al, JAMA, July 9, 2003
- Vol 290, no 2: 252-255. (published after the trial.)
Breastfeeding in Norway – where did they go right? A Gerrard, British Journal of Midwifery, 2001 May, vol. 9, no. 5, p: 294-5, 297-300, (21 ref), http://www.cinahl.com/cgi-bin/refsvc?jid=1450 ISSN: 0969-4900. This comparative paper between Scotland and Norway, analyses the historical, social and cultural factors that influence the prevalence of breast-feeding. It concludes that the strong cultural norm to breast-feed in Norway is partly because of a more relaxed attitude towards the naked human body, a healthier lifestyle in general but also because strategies to reverse the effects of commercial promotion of formula milk, and inconsistent advice by health professionals were implemented at an early stage of the declining trends.
Do consumer infant feeding publications and products available in physicians' offices protect, promote, and support breastfeeding? Valaitis RK, Sheeshka JD, O'Brien MF. School of Nursing, McMaster University, Hamilton, ON, Canada. J Hum Lact. 1997 Sep;13(3):203-8.
Commercial hospital discharge packs for breastfeeding women (Cochrane review). Donnelly A., Sonwden HM, Renfrew MJ, Woolridge MW. In: The Cochrane Library, Issue 2, 2002 Oxford: Update Software.
The U.S. infant formula industry: is direct-to-consumer advertising unethical or inevitable? Cutler BD, Wright RF. Health Mark Q. 2002;19(3):39-55. T, Until Nestle's entry into the U.S. infant formula market in 1988, there was little direct-to-consumer promotion of infant formula. This article provides a historial background of infant feeding in the United States and looks at how mothers' make their infant formula selection.
Violations of the international code of marketing of breastmilk substitutes: prevalence in four countries. Taylor, A BMJ 1998;316:1117-1122. This study by Anna Taylor of the Interagency Group on Breastfeeding Monitoring (IGBM) is based on interviews of 3050 women and 466 health professionals in 165 health facilities in Bangladesh, Poland, South Africa, and Thailand. Prof Lucas dismissed the whole study as unsound. For more information about IGBM see http://www.scfuk.org.uk/development/links/IGBM.htm
It's official: UK recommends exclusive breastfeeding for six months
On May 12th, at the start of National Breastfeeding Week, the UK Department of Health issued a Press Release announcing its decision to recommend exclusive breastfeeding for six months followed by continued breastfeeding with adequate complementary foods, endorsing the World Health Assembly Resolution, (WHA Res 55.25) originally proposed by Brazil. The news, announced by Hazel Blears, (the Health Minister at the time) was greeted with delight by those working to protect breastfeeding and infant health.
In its press statement, the Department of Health pulled no punches about benefits of breastfeeding, stating that:
“there are proven health benefits to breastfeeding for both child and mother in the short and long term. Babies who are breastfed have a lower risk of gastro-enteritis and respiratory and ear infections. There is some evidence that long term breastfeeding may help mothers lose the excess weight they gain during pregnancy and children who are breastfed may be at lower risk of becoming obese later in childhood. Also the risk of pre-menopausal breast cancer in mothers is reduced the longer they breastfeed.”
The announcement generated over 500 news articles, the majority of them positive. The mother and baby press - which receives income from baby food advertising - continues to run adverts promoting complementary foods for use before 6 months. We wait to see if it will oppose the Government’s policy.
The
evidence for the WHO’s recommendations on infant
feeding, including the 6 months exclusive breastfeeding issue,
complementary feeding, emergencies and HIV are availble on
WHO’s website: The Department of Health website has many pages on breastfeeding and even a link to the IBFAN website! |
Baby Feeding Law Group
The announcement was welcomed by professional bodies such as the Royal College of Midwives and the Community Practitioners and Health Visitors’ Association who are both members of the Baby Feeding Law Group. Since its formation in 1997, BFLG has been calling on the Government to adopt the International Code and Resolutions and to protect parents’ rights to a health care system free from commercial pressure. See http://www.babyfeedinglawgroup.org.uk/
Parents Jury
A small survey of 120 mothers by The Parent’s Jury (set up by the Food Commission), found that good intentions to breastfeed were undermined by industry promotion tactics and bad advice from health workers. 76% of the mothers received Bounty Packs when their babies were four months old, which included baby food and follow on milk samples and vouchers. Wyeth/SMA was reported giving a presentation on 'emotional aspects of parenting' at an ante-natal class, offering branded gifts such as pens and writing pads. 25 parents were encouraged by health workers to supplement with formula milk when problems arose. See: http://www.parentsjury.org/
Soya Milk warning
The report on phytoestrogens by the UK Government’s advisory Committee on Toxiticity in Food (COT), outlined the potential risks of soya-based infant formulae. The Scientific Advisory Committee on Nutrition (SACN) also looked at the issue and concluded that there is no clinical need for soya-based formulae. Baby Milk Action is asking the Government why these formulae are still freely available for sale on shelves, with no warnings and without even the need for a prescription. The Food Standards Agency has a web-based Q&A on healthy eating. This explains some of the concerns along with a rather weak warning suggesting that hydrolysed protein infant formulas are better nutritionally than soya-based formulas for allergic babies. FSA suggests consulting a health worker and says it has asked the Dept. of Health to review its advice.
See: http://www.foodstandards.gov.uk/healthiereating/asktheexpert/childbabies/
http://www.soyonlineservice.co.nz/.
Artificial
baby milks: how safe is soya, Tessa Martyn, Vol 6,
No 5 May 2003, Midwives.
India puts health before profits
On 6th May both the houses of the Parliament of India took a historic action to approve a strengthening of 1992 Indian legislation covering baby milk and food marketing. With the passing of the Amendment Bill 2003, baby food manufacturers in India will no longer be permitted to promote products for consumption below the age of two or fund meetings, conferences or any other activities of ‘Health Workers’ and their ’Associations’.
The malnutrition and mortality linked to aggressive promotion is recognized as a major public health problem in India. The 1992 Act itself was a breakthrough in consumer protection, but it left loopholes, which were continuously exploited by babyfood manufacturers.
In 1995, when Nestlé was served with criminal charges over its violation of the Act, the company issued a Writ Petition against the Government, calling for many of the protective clauses to be removed. The fact that the Government has faced up to this challenge, strengthening rather than weakening the law, sends an important signal around the world, at a time when the pressure is being stepped up to deregulate marketing.
Says Dr. Arun Gupta, the National Coordinator of the Breastfeeding Promotion Network of India (BPNI), the IBFAN group working to promote breastfeeding,
"Today, one more battle has been won in the field of protecting, promoting and supporting breastfeeding in India. The Amended Bill strongly directs that infant foods should only be sold for consumption by babies who are more than six months of age and promotion of infant milk substitutes or infant foods is now prohibited for babies up to the age of two years. This comes in support of World Health Assembly’s Resolution 54.2 that defines the period of exclusive breastfeeding to be first six months. It removes any ambiguity whatsoever about the fact that complementary foods should only be introduced after age of six months along with continued breastfeeding for two years or beyond."
A major change is removal of commercial influence on infant feeding. The Indian Delegate to the World Health Assembly in May 2002 made an important intervention during the discussion on WHA Resolution 55.25. He said,
"Commercial enterprises, by definition, are profit-driven entities. It is neither appropriate nor realistic for the WHO to expect that commercial groups will work along with governments and other groups to protect, promote and support breastfeeding."
For more information see: http://www.bpni.org/.
This type of babyfood promotion, which appears in magazines and newspapers all over India, will be illegal under the amendment of the Indian law. |
|
What the new law doesIn addition to the prohibitions contained in the 1992 Act, the Amended Bill 2003, will prohibit the following:
|
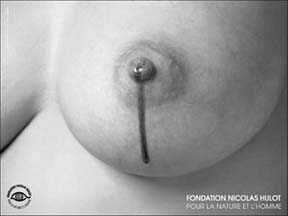
Two ways to campaign Right: The poster - part sponsored by L’Oreal/Nestlé - which appeared all over France in July and has saddened health advocates.
|
|
 |
|
A poster campaign which appeared all over Paris and other French cities in July, just before World Breastfeeding Week, has outraged and saddened health advocates. The poster shows a breast dribbling a dirty, oily fluid - alongside the name of a private foundation which is run by the French TV journalist and green campaigner, Nicolas Hulot. The website of the foundation - http://www.planete-nature.org/ - is part sponsored by L’Oreal, which is part owned by Nestlé. IBFAN, LLL and many other groups have written to Nicolas Hulot, but, since the posters were already in place, the damage had been done.
The failure of Mr Hulot to consult widely before running with this campaign, contrasts with the British environmental groups, Greenpeace, Friends of the Earth, the Women’s Environmental Network and the World Wildlife Fund, who, in December 2002 spent a day with Baby Milk Action, the National Childbirth Trust and others to discuss tactics for a joint campaign to call for phasing out - wherever feasible - of harmful chemicals which build up in the body. There is strong opposition from the chemical industry to such controls, so all the groups were aware of the importance of engaging the public as much as possible in the campaign. The idea of focussing on breastmilk was discussed, but thrown out. The groups realised that such a campaign might well backfire, resulting in women being frightened away from breastfeeding and undermining their children’s health. The WWF advertisement above is an example of how this campaign can be handled.
The EU Commission has launched a European Environment and Health Strategy.
Chemical contamination - what are the concerns?If we tested every infant born today, anywhere in the world, he/she would have a body burden of toxic chemicals which will have been passed from parent to child even before birth. Tiny doses of these chemicals can have a dramatic effect on the developing child, damaging the immune and nervous systems. The chemicals not only cross the placenta, they are found in fatty body secretions such as breastmilk, semen and even ear wax. Campaigns to reduce dioxins and Polychorinated Biphenyls (PCBs) have - over the years - reduced the levels of harmful chemicals found in these body secretions. However, new technologies are creating new chemicals which build up in the body and show up in a similar way. Flame retardants (a group of 70 chemicals) anti-bacterials (Triclosan) and artificial fragrances can all act as hormone disrupters and can be cancer related. Some of these new chemicals have chemical properties similar to PCBs. If levels of contamination are allowed to increase unchecked, breastmilk could become more contaminated. But in this case the health of the unborn child would be already severely compromised and the immune protection and other benefits provided by breastfeeding would be even more important. Breastfeeding, even in a contaminated environment, has a positive impact on development, building a stronger immune system, and counteracting many of the longer-term adverse developmental effects of the pollutants. Artificial baby milks are more contaminated than breastmilk, in different ways. They have been found to contain phthalates, bisphenol A, aluminium and heavy metals, GM ingredients, phytoestrogens and spore bacteria. Any increase in artificial feeding would result in greater contamination of the environment. Responsible NGOs working to reduce environmental pollution focus on the industrial sources of contamination - not on breastfeeding. For action ideas see: http://www.foe.co.uk/campaigns/safer_chemicals/ http://www.wwf.org.uk/chemicals/problem.asp http://www.ibfan.org/english/activities/contaminants/contaminants00.html |
Are the companies moving on 6 months?
Nestlé’s UK Head of Corporate Affairs has written to Baby Milk Action claiming it has changed the labels of its complementary food labels to comply with the requirement that these are not marketed for use before 6 months of age and that the company is ‘leading the way.’
Unfortunately we have yet to see evidence that this is a universal
policy.
UNICEF Hong Kong wrote to Nestlé on 29 May 2003:
“We were pleased to note from the headlines of your International Code Action Report that Nestlé is taking the initiative on 6-month labelling. It was therefore with some disappointment to see that Nestlé Hong Kong is still promoting complementary food from 4 months onwards... the Department of Health of the Hong Kong Government has followed the WHO recommendation of exclusive breastfeeding for the first 6 months of life.”
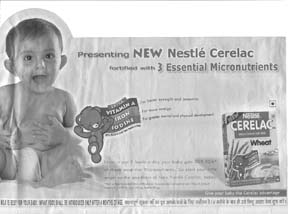
In India, Nestlé’s new labels appeared just as the new legislation was passed in May 2003. However, in Nestlé’s new advertisements the label is covered up and the reference to 6 months is in small print. The new law bans adverts such as this and all promotion for foods for infants under 2 years of age, so the adverts in themselves demonstrate bad faith. The IBFAN group in Bulgaria reports a June 2003 magazine (right) in which Nestlé promotes Sinlac for use from 4 months of age. This says: “Sinlac Baby Menu is a cereal for dietary uses with plant proteins, without gluten, lactose and milk proteins. For every baby over 4 months". |
|
In Lithuania, in June 2003, Nestlé was promoting foods from 4 months in a prize lottery run through the major supermarkets.
Heinz stalls
Misleading though Nestlé's promise is, Baby Milk Action has been using it to pressure other companies to pledge to stop promoting complementary foods for use before 6 months. We have so far only heard from Heinz, which, in a letter from Nutrition Consultant, Dr. Nigel Dickie, states: "...we are not aware of any new recommendations for those mothers who choose to bottle feed." No distinction has been made by the Department of Health or the World Health Assembly between breastfed and bottle-fed infants. Ignoring the nine-year debate on this issue, Heinz also complained: "...unfortunately the Department of Health's announcement of this new recommendation was made without prior warning or consultation with all interested parties."
Most companies in the UK continue to promote complementary foods for use before 6 months of age. Some Boots juices are promoted for use from a few weeks of age.
The 6 months policy globallySince 1994 when WHA Resolution 47.5 recommended exclusive breastfeeding for ‘about 6 months’ many governments have adopted this policy. But those seeking to protect baby food sales in the 4-6 month period refused to accept this and have demanded more and more evidence. The industry concentrated on the Codex Alimentarious Commission, where the global food standards used as bench marks by the World Trade Organisation are set. Baby Milk Action and its IBFAN partners will be attending the next Codex meeting in Bonn in November where the standards will be discussed. The global consensus achieved by the 2001 Resolution, and the Global Strategy on Infant and Young Child Nutrition a year later, were both important breakthroughs, which are moving even the industrialised countries forward. The EU Commission has indicated support, along with Germany, France, the Netherlands and now the UK. Baby Milk Action was asked by the UK Government for information about other national policies. We forwarded messages from IBFAN partners all over the world who are helping their governments implement and monitor infant feeding policies. 70 countries now have official Government policies recommending 6 months exclusive breastfeeding (expressed either as legislation, Presidential Decree, official statement, letter or as guidance to health workers). In Europe these include: Bosnia, France, Belarus, Bulgaria, Czech Republic, Georgia, Germany, Netherlands and Slovakia. |
News
BMJ study exposes marketing violations
The British Medical Journal (18th January 2003) has published a study on baby food marketing in Togo and Burkina Faso which finds widespread violations by baby food companies Nestlé, Danone and others.
The monitoring was conducted at the end of 1999 and mid 2000. For publication in the British Medical Journal the research paper was scrutinised by experts - a process known as peer-review. Nestlé, which was found distributing free samples of infant formulas Al-110 and Preguigoz and producing labels without required warnings amongst other violations, dismissed the findings as old and questioned why they had not been reported to the company. Nestlé ignores the fact that companies are responsible themselves for ensuring that their activities do not violate the marketing requirements. The scientific study was examining whether they are doing so and was conducted with the agreement of the Health Ministries.
Commercial sponsorship in schools - teachers warned of risks
The National Union of Teachers, representing 50% of British teachers, passed a Resolution at its annual conference in April, saying it will provide advice to teachers who object to fundraising schemes linked to business. The NUT has issued a briefing about this issue which alerts teachers to work done by Sustain and to Baby Milk Action’s education pack, Seeing Through the Spin.
WHO tackles Junk Food
Following a May 2002 mandate from the World Health Assembly, WHO is at last addressing the issue of obesity and other food related, non-communicable diseases - taking on the 3 trillion dollar food industry.
Its Global Strategy on Diet, Physical Activity and Health, is a
population-wide, prevention-based strategy which is being developed
through extensive
consultation. It will be presented to the World Health Assembly in
May 2004.
Baby Milk Action has been representing IBFAN at the consultations
which have been taking place with WHO staff and with the former Director
General, Dr Gro Harlem Brundtland. WHO is stressing the benefits
of
breastfeeding throughout the life cycle. We have been urging WHO
to take care in its relationships with the food industry, avoiding
partnerships
and ensuring transparency. We are working closely with a newly formed
global network, called Alliance for People’s Action on Nutrition,
whose position paper can be downloaded from the responses section
on WHO’s Website - http://www.who.int/hpr/global.strategy.shtm
EU moves on health claims
The European Commission has also issued challenges to the food and drink industry with the proposals announced by the EU Commissioner for Consumer Affairs, David Bryne on 17th July. The proposals aim to ban misleading claims and labels and need to be approved by member states and the European Parliament. Baby Milk Action and its IBFAN partners are calling for health claims directed at infants and young children to be banned. The value of the 7 claims permitted under EU regulations are questioned by a new report by the EU Commission’s Scientific Committee on Food (SCF/CS/NUT/IF/65 Final 18th May 2003).
Send comments by 24 October 2003 calling for health claims on foods for infants and young children to be banned to: Akki Khan, Room 115B, Aviation House, 125 Kingsway, London WC2B 6NH Email: nutritionandhealthclaims@foodstandards.gsi.gov.uk
Codex Trust Fund rejects industry funds
Codex Alimentarious standards have been given great significance since
the creation of the World Trade Organisation as they will be used by
WTO as bench marks in the event of trade disputes. IBFAN has been working
for years to ensure the standards meet the World Health Assembly’s
requirements.
At the Codex meeting in February IBFAN and the International Association
of Consumer Food Organisations (IACFO) presented strong arguments why
the Codex Trust Fund should not accept money from food and other industries.
Our concerns were accepted and the Fund will initially seek money only
from governments.
We stressed the importance of redressing the imbalance that exists
in Codex. At the last meeting in Berlin 71% of developed countries
were represented, but only 18% of developing countries. There were
95 government delegates (43% of participants) and 90 industry delegates.
The majority of industry delegates were on Government delegations.
Claims stalled by the US
The issue of health claims was discussed twice this year at Codex. IBFAN is opposed to health claims in principle, but especially on any foods for infants or young children. In June, draft guidelines which called for advertising to be addressed as well as labelling, were about to be adopted, but were held back because of objections by the United States. India, Singapore and Indonesia, all expressed their concerns about health claims saying that they were not ready to scrutinise their validity. Asia is a target for food manufacturers. The Mother and Baby Magazine shown below (November 2002), from Malaysia, contains 26 full colour pages promoting DHA/AA, prebiotics or other ingredients in milks and foods for infants, young children and pregnant women.
 |
Parenting magazines in Malaysia are packed advertisements making health claims. |
in situ at Womad
Theatre group, in situ, performed at the Reading World of Music and Dance (WOMAD) Festival in July. A breast and a tin put forward the arguments of health campaigners and the baby food industry. The audience participated in a quiz and other parts of the action, keeping adults and children alike gripped, even when it started to rain. in situ are a professional group and have worked with primary and secondary schools. Baby Milk Action’s Reading and Berkshire group ran a stall in the One World tent.

For further information on theatre for school children, contact Pete Arnold on 01480 460912 or see http://www.insitutheatre.co.uk/
Breastfeeding in a Globalised world - the new WABA folder
This theme for World Breastfeeding Week 2003 (1-7 August) provides an opportunity to consider the obstacles as well as the benefits of globalisation as a symbol of peace and justice.
Globalisation is the term often used to describe the process of imposing harmonised rules of free trade and free financial flows throughout the whole world. Powered by large corporations and financial markets, globalisation has become a tool to maximise profits. Deregulation and the privatisation of health care systems often put profits before people. In such an environment the economic interests of corporations often take precedence over the sovereign rights of nations, and the needs of mothers and children are easily jeopardized. This folder gives ideas of how to turn this around and use aspects of globalisation to protect health and strengthen breastfeeding cultures.
Useful networks:
Alliance for a corporate-free UN: http://www.corpwatch.org/
People’s health movement: http://phmovement.org/
Order We the people or we the corporations? by Judith Richter (also available as a free download from http://www.ibfan.org/) and the WABA Folder from Baby Milk Action's on-line Virtual shop.